Legend Biotech Reports Third Quarter 2020 Financial Results
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20201116005237/en/
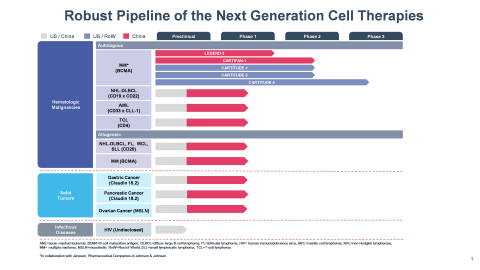
Legend Biotech’s Development Pipeline (Graphic: Business Wire)
“Legend Biotech continues to execute on our corporate strategy, advancing the development of our lead product candidate, ciltacabtagene autoleucel (cilta-cel), in collaboration with
Third Quarter 2020 & Recent Highlights
- On
November 9, 2020 ,Legend Biotech announced thatMs. Ye (Sally) Wang was appointed, effectiveNovember 6, 2020 , as Chairwoman of the Board of Directors ofLegend Biotech . The Board of Directors also named Dr.Ying Huang as CEO ofLegend Biotech , effectiveNovember 6, 2020 .Dr. Huang had been serving as interim CEO sinceSeptember 21, 2020 .Dr. Huang will continue to hold his position as CFO until such time as a successor CFO is identified. - On
November 5, 2020 ,Legend Biotech announced that the Company will present new and updated data from its CARTITUDE-1 and LEGEND-2 studies at the 62ndAmerican Society of Hematology (ASH) Annual Meeting and Exposition taking place virtuallyDecember 5-8, 2020 . - On
September 21, 2020 ,Legend Biotech announced that theCustoms Anti-Smuggling Department of the People’sRepublic of China (“PRC”) had inspected places of business inChina ofLegend Biotech and GenScript Biotech Corporation, Legend Biotech’s majority shareholder, in connection with whatLegend Biotech understands to be an investigation relating to suspected violations of import and export regulations under the laws of the PRC (the “Investigation”) and that Dr. Fangliang Zhang, the Chairman of the Board of Directors and CEO of Legend at that time, had been placed under “residential surveillance” in the PRC. No charges have been filed againstLegend Biotech ,Dr. Zhang , or any of its other officers or directors, and the Company does not believe thatLegend Biotech is a subject of the investigation. - On
August 5, 2020 ,Legend Biotech announced that theChina Center for Drug Evaluation (“CDE”) ofNational Medical Products Administration (“NMPA”) recommended Breakthrough Therapy Designation (“BTD”) for cilta-cel for the treatment of adults with relapsed/refractory multiple myeloma. The designation was granted onAugust 13, 2020 , making cilta-cel the first investigational product to obtain BTD inChina .
Key Upcoming Milestones
- On
Saturday, December 5, 2020 , during the Myeloma session at ASH entitled: Myeloma/Amyloidosis: Therapy, excluding Transplantation: Novel Therapies Targeting B Cell Maturation Antigen in Relapsed/Refractory Multiple Myeloma, the Phase 1b/2 clinical efficacy and safety data from the CARTITUDE-1 study will be presented. - During the ASH Annual Meeting and Exposition, LEGEND-2 data in patients with relapsed or refractory multiple myeloma and extramedullary disease will be presented as a poster.
- Legend Biotech’s collaboration partner
Janssen Biotech, Inc. anticipates initiating the BLA submission for cilta-cel to theU.S. Food and Drug Administration (“FDA”) by the end of 2020 and submitting a marketing authorization application to theEuropean Medicines Agency (“EMA”) in early 2021. Legend Biotech expects to use data from CARTIFAN-1 study to file a regulatory submission inChina in 2021.- Please see Legend Biotech’s comprehensive development pipeline as shown below.
Development Pipeline
The extent to which the COVID-19 pandemic may impact our business and clinical trials is highly uncertain and cannot be predicted with confidence, such as the ultimate geographic spread of the disease, the duration of the outbreak and social distancing regulations, travel restrictions, business closures or business disruptions and the effectiveness of actions taken in
Financial Results for the Three-month and Nine-month Periods Ended
Cash and Cash Equivalents:
As of
Revenue
Revenue for the three months ended
Research and Development Expenses
Research and development expenses for the three months ended
Administrative Expenses
Administrative expenses for the three months ended
Selling and Distribution Expenses
Selling and distribution expenses for the three months ended
Other Income and Gains
Other income and gains for the three months ended
Other Expenses
Other expenses for the three months ended
Loss for the Period
For the three months ended
Grant of restricted share units and share options
On
About
We are engaged in a strategic collaboration with
Cautionary Note Regarding Forward-Looking Statements
Statements in this press release about future expectations, plans and prospects, as well as any other statements regarding matters that are not historical facts, constitute “forward-looking statements” within the meaning of The Private Securities Litigation Reform Act of 1995. These statements include, but are not limited to, statements relating to Legend Biotech’s strategies and objectives; the anticipated timing of, and ability to progress, clinical trials; the ability to make, and the timing of, regulatory submissions globally, including the BLA for cilta-cel to the
|
UNAUDITED INTERIM CONDENSED CONSOLIDATED STATEMENTS OF PROFIT OR LOSS FOR THE THREE AND NINE MONTHS ENDED |
|||||||||||||
|
Three months ended |
Nine months ended |
||||||||||||
|
(in thousands, US$, except per share data) |
2020 |
2019 |
2020 |
2019 |
|||||||||
|
REVENUE |
11,747 |
|
17,674 |
|
34,893 |
|
37,814 |
|
|||||
|
Other income and gains |
1,519 |
|
2,987 |
|
5,315 |
|
6,649 |
|
|||||
|
Research and development expenses |
(63,656 |
) |
(41,917 |
) |
(165,226 |
) |
(95,846 |
) |
|||||
|
Administrative expenses |
(6,038 |
) |
(1,992 |
) |
(13,976 |
) |
(4,704 |
) |
|||||
|
Selling and distribution expenses |
(9,287 |
) |
(4,460 |
) |
(25,389 |
) |
(12,246 |
) |
|||||
|
Other expenses |
(1,249 |
) |
(2 |
) |
(1,331 |
) |
(216 |
) |
|||||
|
Fair value loss of convertible redeemable preferred shares |
— |
|
— |
|
(79,984 |
) |
— |
|
|||||
|
Finance costs |
(90 |
) |
(82 |
) |
(4,169 |
) |
(139 |
) |
|||||
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|||||||||
|
LOSS BEFORE TAX |
(67,054 |
) |
(27,792 |
) |
(249,867 |
) |
(68,688 |
) |
|||||
|
Income taxes credits / (expenses) |
508 |
|
(5 |
) |
4,217 |
|
(341 |
) |
|||||
|
LOSS FOR THE PERIOD |
(66,546 |
) |
(27,797 |
) |
(245,650 |
) |
(69,029 |
) |
|||||
|
Attributable to: |
|||||||||||||
|
Equity holders of the parent |
(66,546 |
) |
(27,797 |
) |
(245,650 |
) |
(69,029 |
) |
|||||
|
LOSS PER SHARE ATTRIBUTABLE TO ORDINARY EQUITY HOLDERS OF THE PARENT |
|||||||||||||
|
Ordinary shares—basic |
(0.25 |
) |
(0.14 |
) |
(1.08 |
) |
(0.35 |
) |
|||||
|
Ordinary shares—diluted |
(0.25 |
) |
(0.14 |
) |
(1.08 |
) |
(0.35 |
) |
|||||
|
ORDINARY SHARES USED IN LOSS PER SHARE COMPUTATION |
|||||||||||||
|
Ordinary shares—basic |
264,328,630 |
|
200,000,000 |
|
226,764,437 |
|
200,000,000 |
|
|||||
|
Ordinary shares—diluted |
264,328,630 |
|
200,000,000 |
|
226,764,437 |
|
200,000,000 |
|
|||||
|
UNAUDITED INTERIM CONDENSED CONSOLIDATED STATEMENTS OF FINANCIAL POSITION AS AT |
|||||
|
|
|
||||
|
(in thousands, US$) |
|||||
|
NON-CURRENT ASSETS |
|||||
|
Property, plant and equipment |
96,125 |
70,079 |
|
||
|
Advance payments for property, plant and equipment |
675 |
665 |
|
||
|
Right-of-use assets |
8,077 |
9,348 |
|
||
|
Intangible assets |
1,042 |
519 |
|
||
|
Total non-current assets |
105,919 |
80,611 |
|
||
|
CURRENT ASSETS |
|||||
|
Inventories |
1,513 |
1,157 |
|
||
|
Trade receivables |
— |
29,991 |
|
||
|
Prepayments, other receivables and other assets |
24,662 |
16,777 |
|
||
|
Financial assets at fair value through profit or loss |
1,175 |
— |
|
||
|
Pledged short-term deposits |
430 |
256 |
|
||
|
Time deposits |
125,559 |
75,559 |
|
||
|
Cash and cash equivalents |
449,381 |
83,364 |
|
||
|
Total current assets |
602,720 |
207,104 |
|
||
|
Total assets |
708,639 |
287,715 |
|
||
|
CURRENT LIABILITIES |
|||||
|
Trade and notes payables |
7,399 |
9,586 |
|
||
|
Other payables and accruals |
67,889 |
70,854 |
|
||
|
Lease liabilities |
1,445 |
1,027 |
|
||
|
Contract liabilities |
46,789 |
46,294 |
|
||
|
Total current liabilities |
123,522 |
127,761 |
|
||
|
NON-CURRENT LIABILITIES |
|||||
|
Contract liabilities |
245,641 |
277,765 |
|
||
|
Lease liabilities |
2,543 |
5,058 |
|
||
|
Government grants |
2,033 |
— |
|
||
|
Total non-current liabilities |
250,217 |
282,823 |
|
||
|
Total liabilities |
373,739 |
410,584 |
|
||
|
EQUITY |
|||||
|
Share capital |
26 |
20 |
|
||
|
Reserves / (deficits) |
334,874 |
(122,889 |
) |
||
|
Total ordinary shareholders’ equity / (deficit) |
334,900 |
(122,869 |
) |
||
|
Total equity / (deficit) |
334,900 |
(122,869 |
) |
||
|
Total liabilities and equity |
708,639 |
287,715 |
|
||
|
UNAUDITED INTERIM CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS FOR THE THREE AND NINE MONTHS ENDED |
||||||||||||
|
|
Three months ended |
Nine months ended |
||||||||||
|
(in thousands, US$) |
2020 |
2019 |
2020 |
2019 |
||||||||
|
LOSS BEFORE TAX |
(67,054 |
) |
(27,792 |
) |
(249,867 |
) |
(68,688 |
) |
||||
|
CASH FLOWS (USED IN)/GENERATED FROM OPERATING ACTIVITIES |
(64,375 |
) |
19,947 |
|
(167,056 |
) |
(23,078 |
) |
||||
|
CASH FLOWS USED IN INVESTING ACTIVITIES |
(58,623 |
) |
(21,194 |
) |
(85,334 |
) |
(172,103 |
) |
||||
|
CASH FLOWS FROM/(USED IN) FINANCING ACTIVITIES |
9,663 |
|
(154 |
) |
618,221 |
|
21,346 |
|
||||
|
|
(113,335 |
) |
(1,401 |
) |
365,831 |
|
(173,835 |
) |
||||
|
Effect of foreign exchange rate changes, net |
325 |
|
(44 |
) |
186 |
|
(55 |
) |
||||
|
Cash and cash equivalents at beginning of the period |
562,391 |
|
37,721 |
|
83,364 |
|
210,166 |
|
||||
|
CASH AND CASH EQUIVALENTS AT END OF THE PERIOD |
449,381 |
|
36,276 |
|
449,381 |
|
36,276 |
|
||||
|
ANALYSIS OF BALANCES OF CASH AND CASH EQUIVALENTS |
||||||||||||
|
Cash and bank balances |
575,370 |
|
147,592 |
|
575,370 |
|
147,592 |
|
||||
|
Less: Pledged short-term deposits |
430 |
|
255 |
|
430 |
|
255 |
|
||||
|
Time deposits |
125,559 |
|
111,061 |
|
125,559 |
|
111,061 |
|
||||
|
Cash and cash equivalents as stated in the statement of financial position |
449,381 |
|
36,276 |
|
449,381 |
|
36,276 |
|
||||
|
Cash and cash equivalents as stated in the statement of cash flows |
449,381 |
|
36,276 |
|
449,381 |
|
36,276 |
|||||
View source version on businesswire.com: https://www.businesswire.com/news/home/20201116005237/en/
Media and Investor Relations:
Relations,
investor@legendbiotech.com
media@legendbiotech.com
For Medical Affairs inquiries, please contact:
tonia.nesheiwat@legendbiotech.com or medicalinformation@legendbiotech.com
Source:



